 The conclusions of a new survey regarding quality of life for Ulcerative Colitis patients were recently released online and through social media channels as a means of improving care and treatment of the disease. The survey, which was commissioned and funded by MSD, the UK subsidiary of Merck & Co., Inc., together with Crohn’s and Colitis UK (CCUK), studied 684 people with Ulcerative Colitis (UC) who answered questions regarding the level of disease control, the impact on quality of life, treatment satisfaction, and satisfaction after having a colectomy, a surgical procedure where the colon is removed, partially or completely.
The conclusions of a new survey regarding quality of life for Ulcerative Colitis patients were recently released online and through social media channels as a means of improving care and treatment of the disease. The survey, which was commissioned and funded by MSD, the UK subsidiary of Merck & Co., Inc., together with Crohn’s and Colitis UK (CCUK), studied 684 people with Ulcerative Colitis (UC) who answered questions regarding the level of disease control, the impact on quality of life, treatment satisfaction, and satisfaction after having a colectomy, a surgical procedure where the colon is removed, partially or completely.
The majority of the surveyed patients were women (74%), and the ages varied between less than 24 years old (18%), 25–39 (46%) and 40–54 years of age (29%).
Between the participants, nearly 1 in 10 had undergone a colectomy. These patients reported frequent fatigue (74%), abdominal pain (54%) and diarrhea (33%). While fatigue was reported as the worst symptom to cope with, the most common post-operative complications were related to infections (43%), followed by pouch leakage (31%) and pouchitis (23%), a condition characterized by inflammation of the ileal pouch (an artificial rectum surgically created out of ileal gut tissue in patients who have had a colectomy).
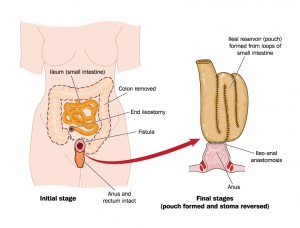 The results of the survey were announced in parallel with the ongoing NICE Multiple Technology Appraisal (MTA) of biologics to treat moderate to severe UC. While current NICE guidelines restrict biologics use in hospitalized severe UC, the survey’s finding suggest the need for a reevaluation of the guidelines to treat moderate-to-severe UC that fails conventional therapies.
The results of the survey were announced in parallel with the ongoing NICE Multiple Technology Appraisal (MTA) of biologics to treat moderate to severe UC. While current NICE guidelines restrict biologics use in hospitalized severe UC, the survey’s finding suggest the need for a reevaluation of the guidelines to treat moderate-to-severe UC that fails conventional therapies.
Chris Probert, Professor of Gastroenterology at the University of Liverpool, Honorary Consultant Gastroenterologist at Royal Liverpool Hospital, chair of the IBD Committee for the British Society of Gastroenterology and vice-chair of the Clinical Advisers Committee for Crohn’s and Colitis UK, commented, “Although many people with ulcerative colitis will respond well to conventional therapies, there will always be a subset of those who fail to respond to these treatments and alternative choices to surgery are needed. Biologics could offer a life-transforming opportunity for these patients, who are often young adults, and may even help delay the need for a colectomy.”
This is particularly important, since 75% of patients surveyed reported dissatisfaction with their current treatment to reduce symptoms and control the disease. Half of patients surveyed reported hospitalization periods of at least 16 days over the last five years.
Helen Terry, Director of Policy and Research at patient charity Crohn’s and Colitis UK, noted, “Biological therapies offer hope to people with ulcerative colitis for whom conventional therapies are failing to control symptoms, not only so that surgery may be avoided, but so they can resume their lives.”


Help please.
We have a son who’s just turned 14 – Will was diagnosed with severe ulcerative pan-colitis 2 years ago. After going through all of the usual drugs he is now on Infliximab which has mainly worked in reducing the inflammation in his colon. He will be on Infliximab indefinitely however our enormous difficulty is that Will suffers with underlying fatigue which does not go away and when combined with severe stomach pain, headaches, a c-diff infection he has hardly attended school in the past 2 years. We tried a ‘drug holiday’ a year ago but the fatigue did not lift and his colon returned to a raging mess – back to Infliximab.
Will doesn’t have quality of life and his specialist has no idea what to do. Is there any one who knows of others suffering the same as our son? Surely Will is not the only person like this?! We desperately need help to get him back into participating in life again.
We live in Adelaide, South Australia. Regards
Sharon
Sharon,
Have you considered in Indigo Naturalis? Conventional UC treatments such as Imuran, Enemas and Mesalazine stopped working for me. I really wanted to avoid infliximab due to the side effects. My doctor sent me to a doctor of combined medicine in Hiroshima ( I live in Japan). I was skeptical of alternative medicine but I decided to give it a go. 6 months later I have stopped all conventional medicine and I take 2 sachets of Indigo Naturalis daily. My bowel movements are back to normal, I have no fatigue and no dietary restrictions. The side effects from immunotherapy have all gone. Indigo Naturalis gave me my life back.
You can buy the herb in Australia quite easily; however, I recommend contacting Hiroshima Sky clinic. They use a special mixture and also monitor my health very closely. They also have many patients in foreign countries and can speak English. Either way I recommend trying Indigo Naturalis. But don’t just take my word for it look at scholarly articles on Qing Dai (Indigo Naturalis) for UC.
Justin