British medical technology firm 11health has announced UK availability of what it claims is the first ever patient-developed medical technology device. The Ostom-i Alert is projected to help the roughly 20,000 people in the UK who have stoma surgery annually address conditions resulting from surgeries performed to address colorectal cancer, bladder cancer, ulcerative colitis and Crohn’s disease.
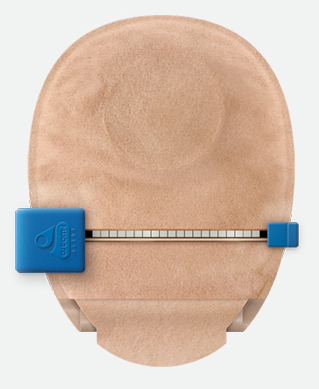
The Ostom-i Alert sensor is a discrete innovative device that alerts patients as to the fullness of their ostomy/stoma bags to help the wearer decide its time to empty them in plenty of time to prevent both fluid and solid leakage, enabling patients to avoid embarrassing situations when out, or in particular when asleep. Patients can also use the device to send output levels automatically to nurses and clinicians.
The Ostom-i Alert is intended to help patients and their families, self-manage their Ostomy. It clips on to any ostomy bag and sends Bluetooth alerts to an app on an iOS or Android smartphone when the bag is getting full to a set point where the user wishes to be notified. The device also captures guidance information about volume of output over a time period and allows users to email that information automatically to nurses and clinicians. In addition, this data is stored safely on 11health’s website, allowing user access it at any given time. Each device is single use only and last up to three months.
The device allows hospitals to remotely monitor the patients through a shared Cloud based portal. This allows potential dehydration and blockage problems to be diagnosed earlier by clinicians, which could reduce both nursing time and hospital re-admission rates. Patients and clinical staff can track and share real-time patient data about the volume of output over specific time periods. This view of a patient’s day-to-day experience will help doctors adjust medication and promote healthy self-management.
11health was founded by Michael Seres, a long-term ostomy patient turned innovator who created the Ostom-i Alert device from his hospital bed. Mr. Seres was diagnosed with the incurable bowel condition Crohns Disease at age 12. After over 20 operations and intestinal failure he became the 11th person to undergo a small bowel transplant in the UK at The Churchill Hospital, Oxford, England. His own experience as an ostomy patient led him to designing the initial Ostom-i prototype whilst recovering from transplant. 11health recently secured Food and Drug Administration (FDA) approval in the US, and Ostom-i Alert has been approved for patient use.
“Our device started life on a hospital transplant ward,” Mr, Seres recalls. “I woke up went to feel my stomach and came across my stoma bag. As part of my bowel transplant I had to have an ileostomy. It took me about 3 weeks to actually look at my stoma and then another week to pluck up the courage to change the bag.
“Once that was accomplished I got used to emptying and changing by bag/pouch. However I soon realized that it wasn’t quite as simple as pressing a call bell and asking a nurse to help. Output would occur without much notice and that led to leaks and even overflows — especially at night. In addition my transplant team wanted me to track the volume of output and the timing. All of this led to 2am showers due to overflows and emptying my bag into measuring jugs.”
 “There had to be an alternative way except I could not find it. So with the help of the Internet I started buying a few spare parts and working out how I could improve my own life. That was how Ostom-iwas conceived and a prototype developed. From the hospital bed to this point has taken a great deal of hard work but I am proud of what we have achieved. I hope that our device helps make your life as an ostomy patient that little bit easier.”
“There had to be an alternative way except I could not find it. So with the help of the Internet I started buying a few spare parts and working out how I could improve my own life. That was how Ostom-iwas conceived and a prototype developed. From the hospital bed to this point has taken a great deal of hard work but I am proud of what we have achieved. I hope that our device helps make your life as an ostomy patient that little bit easier.”
A published author and professional speaker, aside from being managing director of 11health, Michael Seres is the recipient of Caregiver/Patient Duo Scholarship at the 2012 Stanford Medicine X Conference, and develops patient engagement strategies for hospitals, primary care trusts and patient groups. He has a background in consumer product licensing and merchandising having worked with major brands such as Fifa, UEFA, The X Factor and The Apprentice. He is the patient lead for the main UK health twitterchat #NHSSM & digital strategy advisor to The Oxford Transplant Foundation where he helped implement the first skype clinics.
Michael Seres observes: “Being a patient isn’t easy, especially for those who suffer from long term chronic bowel conditions, like myself. Given the increasing pressures faced by healthcare systems and staff globally, I couldn’t believe that there was no simple technology device to manage the accidents and embarrassment of overflowing bags for patients, and save time for clinical teams whilst providing real-time patient data to monitor outpatient progress.”
He continues, “Patient self-management is a growth area, as healthcare systems buckle under the weight of people living longer and rising hospital costs. With the availability of the Ostom-i Alert, bowel patients will no longer suffer from unnecessary accidents and will be able to live as normal lives as possible. The device, developed ‘by patients for patients’, sends information to health professionals to simplify patient monitoring, and we estimate it can save 33% of current NHS stoma care costs.”
Georgios Vrakas, Consultant Transplant Surgeon, Small Bowel Transplantation Oxford University Hospital Trust, comments regarding Ostom-i Alert: “This product is inspirational. Something so simple in concept, yet so incredibly useful. This was tricky to invent, but through sheer determination Michael has created something that should become standard issue for every ostomy patient. There is no question that it will be life changing for millions of bowel disease sufferers.”
The Ostom-i Alert device, for which 11health has obtained a patent, is manufactured in Lancaster (UK) and Utah (USA). The device can be purchased online and costs about £75.00 per unit. Patients can set individual alerts as to when they wish to be notified. Each device lasts up to three months.
Ostom-i 24 Hour Help Line
US Toll Free Number
1-866-834-8356
UK Free Phone Number
+44 0843 320 5126
Sources:
11health
Image Credits:
11health
